Leukopaks
From Europe. For Europe.
Leukapheresis, cryopreservation and cell products from Vienna. 500+ qualified donors, GMP-compliant processes, worldwide shipping.
Leukapheresis
Gentle extraction of leukocytes from the circulation of healthy donors and patients.
Learn moreCryopreservation
Professional cryopreservation, long-term storage, and temperature-controlled shipping — ensuring your cell products reach their destination safely.
Learn moreLogistics
Time-critical transport of highly sensitive cell products — with a validated cold chain, GDP-compliant processes, and personal supervision of every shipment.
Learn moreOur USP: Flexibility & Speed
4 days leadtime
Offers one of the shortest in Europe for non-mobilized leukapheresis.
6–16h EU, 24h worldwide
Delivery time within the EU — every shipment to date delivered within 24 hours worldwide.
Maximum flexibility
Fast, adaptable processes specifically tailored to the requirements of time-sensitive cell therapy programs.
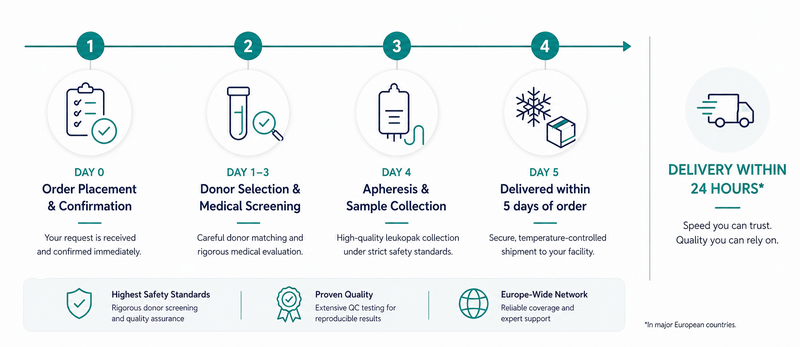
Our Process
From request to delivery in 5 days
From first contact to temperature-controlled delivery — our process is designed to meet your clinical and R&D timelines.
We handle definition of product specifics, donor selection, scheduling, collection, documentation and logistics coordination as your single point of contact — from first inquiry to courier pickup.
More about our processCells for Therapy & Research
Our cell products are used across a wide range of medical and scientific fields.
CAR-T Cell Therapy
CAR-T cell therapy is revolutionizing the treatment of blood cancer. CytoCare provides the critical starting material — high-quality leukapheresis products that serve as the basis for genetic modification of T cells.
Learn moreDendritic Cell Therapy
Dendritic cells are the key players of the immune system. Monocytes derived from our leukapheresis products are differentiated into dendritic cells and used for personalized immunotherapies.
Learn moreResearch & Pharma
For biomedical research, drug discovery, and process development, we supply fresh and cryopreserved cell products of the highest quality — reliable, reproducible, and GMP-compliant.
Learn moreFrom Inquiry to Delivery

Quality
Highest Standards for Your Safety
Our quality management system ensures compliance with all relevant regulations and standards.
From donor selection through collection to delivery — every step is subject to rigorous quality controls. Our processes are GMP-compliant and regularly audited.
Quality Management